Research suggests anti-inflammatory protein may trigger plaque in Alzheimer’s disease
Inflammation has long been studied in Alzheimer’s, but in a counterintuitive finding reported in a new paper, University of Florida researchers have uncovered the mechanism by which anti-inflammatory processes may trigger the disease.
This anti-inflammatory process might actually trigger the build-up of sticky clumps of protein that form plaques in the brain. These plaques block brain cells’ ability to communicate and are a well-known characteristic of the illness.
The finding suggests that Alzheimer’s treatments might need to be tailored to patients depending on which forms of Apolipoprotein E, a major risk factor for Alzheimer’s disease, these patients carry in their genes.
The researchers have shown that the anti-inflammatory protein interleukin 10, or IL-10, can actually increase the amount of apolipoprotein E, or APOE, protein – and thereby plaque – that accumulates in the brain of a mouse model of Alzheimer’s, according to the study, published online today (Jan. 22) in the journal Neuron.
In the 1990s, researchers theorized that using nonsteroidal anti-inflammatory drugs, or NSAIDs, might protect people from the onset of Alzheimer’s by dampening inflammation that released a cascade of harmful proteins. Though NSAIDs were shown to be effective in some studies, other research that evaluated a group of participants taking NSAIDs over time failed to show any clear protective benefit.
“There are many different kinds of NSAIDs,” said Todd Golde, M.D., Ph.D., director of the Center for Translational Research in Neurodegenerative Disease and the paper’s lead author. “Not all NSAIDs are equal, and it wasn’t clear what else they were doing when they were addressing their intended target.”
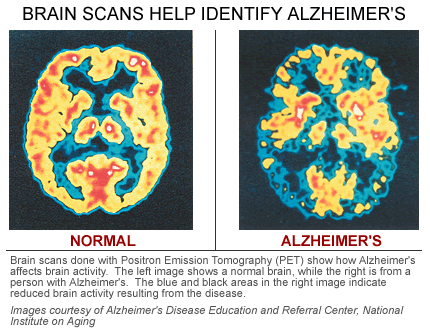
Scientists are not absolutely sure what causes Alzheimer’s but plaques and tangles are prime suspects in cell death and tissue loss in the Alzheimer brain.
Plaques are abnormal clusters of chemically “sticky” proteins called beta-amyloid that build up between nerve cells. The most damaging form of beta-amyloid may be groups of a few pieces rather than the plaques themselves. The small clumps may block cell-to-cell signaling at synapses. They may also activate immune system cells that trigger inflammation and devour disabled cells.
Tangles form inside dying cells. Tangles are twisted fibers of a protein called tau. In healthy areas, tau helps keep the transport system on track. But in areas where tangles are forming, the twisted strands of tau essentially disintegrate the transport system so that nutrients and other essential supplies can no longer move through the cells, which eventually die.
Though most people develop some plaques and tangles as they age, those with Alzheimer’s tend to develop far more. The plaques and tangles tend to form in a predictable pattern, beginning in areas important in learning and memory and then spreading to other regions.
Plaques form when protein pieces called beta-amyloid (BAY-tuh AM-uh-loyd) clump together. Beta-amyloid comes from a larger protein found in the fatty membrane surrounding nerve cells.
Beta-amyloid is chemically “sticky” and gradually builds up into plaques.
The most damaging form of beta-amyloid may be groups of a few pieces rather than the plaques themselves. The small clumps may block cell-to-cell signaling at synapses. They may also activate immune system cells that trigger inflammation and devour disabled cells.
 Previously, researchers hypothesized that a flood of proteins, called cytokines, involved in promoting inflammation in the brain contributed to the formation of plaque in Alzheimer’s disease. However, in this publication, the UF researchers provide new evidence that anti-inflammatory stimuli may actually increase plaque.
Previously, researchers hypothesized that a flood of proteins, called cytokines, involved in promoting inflammation in the brain contributed to the formation of plaque in Alzheimer’s disease. However, in this publication, the UF researchers provide new evidence that anti-inflammatory stimuli may actually increase plaque.
“This is another piece of evidence that overturns the long-held hypothesis that a ‘cytokine storm’ creates a self-reinforcing, neurotoxic feedback loop that promotes amyloid-beta (plaque) deposition,” said Paramita Chakrabarty, Ph.D., a member of the UF Center for Translational Research in Neurodegenerative Disease, an assistant professor in the UF College of Medicine department of neuroscience and the paper’s co-author.
The researchers said that a person’s risk of developing Alzheimer’s hinges on the relationship between IL-10 and APOE. APOE clears the cell of many different proteins, including the protein amyloid-beta, which contributes to the buildup of plaque. But there are several different forms of APOE in cells, which differ from each other by only one or two amino acids. The form called APOE4 is the largest known genetic risk factor in Alzheimer’s disease, while APOE2 is thought to be protective, Golde said.
Amyloid Plaques
One of the hallmarks of Alzheimer’s disease is the accumulation of amyloid plaques between nerve cells (neurons) in the brain. Amyloid is a general term for protein fragments that the body produces normally. Beta amyloid is a protein fragment snipped from an amyloid precursor protein (APP). In a healthy brain, these protein fragments are broken down and eliminated. In Alzheimer’s disease, the fragments accumulate to form hard, insoluble plaques.
Neurofibrillary Tangles
Neurofibrillary tangles are insoluble twisted fibers found inside the brain’s cells. These tangles consist primarily of a protein called tau, which forms part of a structure called a microtubule. The microtubule helps transport nutrients and other important substances from one part of the nerve cell to another. In Alzheimer’s disease, however, the tau protein is abnormal and the microtubule structures collapse.
“About 15 to 17 percent of the population has the APOE 4 allele, and about 50 percent of people with Alzheimer’s have it,” Golde said.
 In this case, the authors showed that the anti-inflammatory protein IL-10 actually increases levels of all types of mouse APOE, which resembles human APOE. In the mouse model, APOE binds with amyloid-beta rather than clearing it from the brain, accelerating buildup of plaque in the brain of a mouse with Alzheimer’s. How an anti-inflammatory therapy based on IL-10 expression might alter risk for Alzheimer’s may depend on the genetic variant of APOE protein the person is carrying. If the person has an APOE4 allele the researchers predict the risk for Alzheimer’s would increase.
In this case, the authors showed that the anti-inflammatory protein IL-10 actually increases levels of all types of mouse APOE, which resembles human APOE. In the mouse model, APOE binds with amyloid-beta rather than clearing it from the brain, accelerating buildup of plaque in the brain of a mouse with Alzheimer’s. How an anti-inflammatory therapy based on IL-10 expression might alter risk for Alzheimer’s may depend on the genetic variant of APOE protein the person is carrying. If the person has an APOE4 allele the researchers predict the risk for Alzheimer’s would increase.
“In one way, this study offers additional insight into how environmental influences interacts with people’s underlying genotypes to alter their risk for diseases,” Golde said. “We know that people are exposed to various inflammatory or anti-inflammatory stimuli throughout their lives. Depending on what their genotype is, that exposure may in some cases protect them from Alzheimer’s, or, in other cases, increase their risk for Alzheimer’s.”
###
The research was funded in part through an $8.4 million grant to speed up the process of finding therapies for Alzheimer’s disease from the National Institutes of Health’s Office of the Director, with additional funding from the National Institute on Aging and the Ellison Medical Foundation. Next, the researchers plan to carry out more thorough and mechanistic studies to exactly understand how an increase in APOE protein induced by IL-10 will affect amyloid plaque deposition in mice carrying different alleles of human APOE.
###
Morgan Sherburne
.(JavaScript must be enabled to view this email address)
352-273-6160
University of Florida
Journal
Neuron
Funder
NIH/Office of the Director, NIH/National Institute on Aging, Ellison Medical Foundation





